Properties of fluids Case Study
Introduction
A fluid is a substance that deforms continuously under the action of a shear force. A fluid may be a liquid or a gas; it offers resistance to a change of shape and is capable of flowing. Liquid and gas are distinguished as follows:
• A gas completely fills the space in which it is contained; a liquid usually has a free surface.
• A gas is a fluid which can be compressed relatively easily and is often treated as such; a liquid can be compressed only with difficulty
Dimension And Units
Dimension = A dimension is the measure by which a physical variable is expressed quantitatively. Unit = A unit is a particular way of attaching a number to the quantitative dimension. Thus length is a dimension associated with such variables as distance, displacement, width, deflection, and height, while centimeters or meters (or inchs etc) are both numerical units for expressing length.
Properties of Fluids
Properties of fluids determine how fluids can be used in engineering and technology. They also determine the behaviour of fluids in fluid mechanics. The following are some of the important basic properties of fluids:
pressure, temperature, density, specific weight, viscosity, specific heat, internal energy, bulk modulus of elasticity and compressibility, velocity of sound, equations of state
International System of Units (SI):
1 Pa = 1 N/m² ; 1 bar = 10⁵ Pa . N = kgm/s²
Kinds of pressure
Fluid fow pressure = static presure,
Dynamic pressure,
Total pressure,
Absolute pressure,
Atmospheric pressure,
Gauge pressure,
Vacuum,
Hydrostatic pressure etc
Temperature:
It is the property that determines the degree of hotness or coldness or the level of heat intensity of a fluid. Temperature is measured by using temperature scales.There are 3 commonly used temperature scales. They are
- Celsius (or centigrade) scale
- Fahrenheit scale
- Kelvin scale (or absolute temperature scale)
Specific density :
In general, density of a fluid decreases with increase in temperature. It increases with increase in pressure.
The ideal gas equation is given by:
Note: The density of standard liquid (water) is 1000 kg/m³.
Specific Gravity:
Specific gravity is the ratio of specific weight of the given fluid to the specific weight of standard fluid. It is denoted by the letter ‘S’. It has no unit.
Specific Volume :
Specific volume of a fluid is defined as the volume of a fluid occupied by a unit mass or volume per unit mass of a fluid.
Thus, specific volume is the reciprocal of mass density. It is expressed as m³/kg. It is commonly applied to gases.
Newton’s law of viscosity
In a liquid, viscosity decreases with increase in temperature. In a gas, viscosity increases with increase in temperature.
compressible fluid
A compressible fluid is a fluid in which significant density variations that occur during its flow have to be considered, as is usually the case with vapors and gases.
Introduction
A fluid is a substance that deforms continuously under the action of a shear force. A fluid may be a liquid or a gas; it offers resistance to a change of shape and is capable of flowing. Liquid and gas are distinguished as follows:
• A gas completely fills the space in which it is contained; a liquid usually has a free surface.
• A gas is a fluid which can be compressed relatively easily and is often treated as such; a liquid can be compressed only with difficulty
Dimension = A dimension is the measure by which a physical variable is expressed quantitatively. Unit = A unit is a particular way of attaching a number to the quantitative dimension. Thus length is a dimension associated with such variables as distance, displacement, width, deflection, and height, while centimeters or meters (or inchs etc) are both numerical units for expressing length.
Properties of Fluids
Properties of fluids determine how fluids can be used in engineering and technology. They also determine the behaviour of fluids in fluid mechanics. The following are some of the important basic properties of fluids:
pressure, temperature, density, specific weight, viscosity, specific heat, internal energy, bulk modulus of elasticity and compressibility, velocity of sound, equations of state
Pressure:
Property defined as force per unit area (figure 1):
P= Fp/A
Fp - force applied on a surface A in a direction perpendicular to that surface
 |
| FIG 1 |
1 Pa = 1 N/m² ; 1 bar = 10⁵ Pa . N = kgm/s²
Kinds of pressure
Fluid fow pressure = static presure,
Dynamic pressure,
Total pressure,
Absolute pressure,
Atmospheric pressure,
Gauge pressure,
Vacuum,
Hydrostatic pressure etc
Temperature:
It is the property that determines the degree of hotness or coldness or the level of heat intensity of a fluid. Temperature is measured by using temperature scales.There are 3 commonly used temperature scales. They are
- Celsius (or centigrade) scale
- Fahrenheit scale
- Kelvin scale (or absolute temperature scale)
Symbol: T, t
 |
| FIG 2 |
SI:
K = Kelvin
C⁰ = Degree Celsius
K = 273,16 + [C⁰]
Kinds of temperature :
Fluid flow temperature;
Total (stagnation) temperature etc
Specific density or specific mass or mass density is the mass per unit volume of the fluid.
Density is denoted by the symbol ‘ρ’. Its unit is kg/m³.
ρ = mass/volume = m/v
 |
| FIG 3 |
Units:
SI System: kg/m³
The ideal gas equation is given by:
Note: The density of standard liquid (water) is 1000 kg/m³.
Specific weight:
Specific weight or weight density of a fluid is the weight per unit volume
γ =weight / volume= W/V
γ = ρ × g
 |
| FIG 4 |
With increase in temperature volume increase and hence specific weight decreases. With increase in pressure volume decreases and hence specific weight increases.
Therefore specific weight = specific density * acceleration due to gravity
γ = ρ × g
Units:
SI System: N/m³
Specific Gravity:
Specific gravity is the ratio of specific weight of the given fluid to the specific weight of standard fluid. It is denoted by the letter ‘S’. It has no unit.
 |
| FIG 5 |
Specific Volume :
Specific volume of a fluid is defined as the volume of a fluid occupied by a unit mass or volume per unit mass of a fluid.
 |
| FIG 6 |
Thus, specific volume is the reciprocal of mass density. It is expressed as m³/kg. It is commonly applied to gases.
Viscosity:
Viscosity is a measure of the resistance of a fluid to deform under shear stress. It is commonly perceived as "thickness", or resistance to flow.Fig 7. .
Viscosity describes a fluid's internal resistance to flow and may be thought of as a measure of fluid friction.
In general, in any flow, layers move at different velocities and the fluid's viscosity arises from the shear stress between the layers that ultimately opposes any applied force.
 |
| FIG 7 |
a liquid between the fixed plate and the movable plate at a distance ‘Y’ apart, ‘A’ is the contact area (Wetted area) of the movable plate, ‘F’ is the force required to move the plate with a velocity ‘U’ According to Newton’s law shear stress is proportional to shear strain.
 |
| FIG 8 |
τ = µ du / dy
‘µ’ is the constant of proportionality called Dynamic Viscosity or Absolute Viscosity or Coefficient of Viscosity or Viscosity of the fluid.In a liquid, viscosity decreases with increase in temperature. In a gas, viscosity increases with increase in temperature.
μ - Dynamic (absolute) viscosity in Ns/m²; in some literature the symbol η is used (η=μ).
SI units: kg/ms=Ns/m²; P = dyns/cm² = g/cms = 0,1 Ns/m²; cP = 10⁻² P
Kinematic viscosity, ν, is very often used in the hydraulic computations. Kinematic viscosity is defined as ratio of the dynamic viscosity μ and density ρ:
ν = μ / ρ
SI units: m²/s; St = cm²/s = 10-4 m²/s ; cSt = 10⁻² St = 10⁻⁶ m²/s ; mSt = 10⁻³ St
Specific heat, c:
Specific heat capacity, also known simply as specific heat, is the ratio of the quantity of heat flowing into a substance per unit mass to the change in temperatutre
= measure of the heat energy required to increase the temperature of one kg of a substance by one Kelvin.
C= Q/m dT
where :
Q : heat added or removed
dT : change of temperature
m : mass
 |
| FIG 9 |
SI units: J/kgK
Cp= specific heat at constant pressure;
Cv = specific heat at constant volume.
Specific internal energy, u:
Defined as energy per unit mass, due to the kinetic and potential energies bound into the substance by its molecular activity and depends primarily on temperature
For a perfect (ideal) gass:
du = Cv×dt
for Cv = const :
u2-u1=Cv×(T2-T1)
SI units: J/kg
Specific entalpy, h:
Sum of the internal energy and energy due to the pressure change:
Sum of the internal energy and energy due to the pressure change:
P/ρ is the flow energy, also called the flow work which is the energy per unit mass needed to move the fluid and maintain flow.
enthalpy of an ideal gas can be expressed in terms of the specific heats as
du =cv dT and di = cp dT
where cv and cp are the constant-volume and constant-pressure specific heats of the ideal gas.
For incompressible substances, the constant-volume and constant-pressure specific heats are identical. Therefore, Cp = Cv =C for liquids,
For a perfect (ideal) gass:
For Cp const :
SI units: J/kg
Compressibility and Bulk modulus of elasticity:
Fluids usually expand as they are heated or depressurized and contract as they are cooled or pressurized. But the amount of volume change is different for different fluids,
 |
| FIG 12 |
Properties that relate volume changes to the changes in pressure and temperature. Two such properties are the bulk modulus of elasticity k and the coefficient of volume expansion 𝞫
Compressibility β is a measure of the relative volume change of a fluid or solid as a response to a pressure (or mean stress) change:
The bulk modulus of elasticity is defined as reciprocal of compresibility (T = constant):
SI units fo Ev : N/m²
Liquids (incompressible fluids) have large values of Ev
Velocity of sound, c:
 |
| FIG 13 |
In case of isentropic adiabatic process of a gass p/ρ𝜅 = const:
k=Cp/Cv
For liquids c is determined from experimental values of k
Vapor pressure, pv - cavitation presuure, pk:
The vapor pressure Pv of a pure substance is defined as the pressure exerted by its vapor in phase equilibrium with its liquid at a given temperature. Pv is a property of the pure substance, and turns out to be identical to the saturation pressure Psat of the liquid (Pv = Psat).
All solids and liquids have a tendency to evaporate to a gaseous form, and all gases have a tendency to condense back.
At any given temperature, for a particular substance, there is a partial pressure at which the gas of that substance is in dynamic equilibrium with its liquid or solid forms. This is the vapor pressure of that substance at that temperature.
Cavitation = rapid (almost "explosive") change of of phase from liquid to vapor
 |
| FIG 14 |
the liquid pressure in liquid-flow systems dropping below the vapor pressure at some locations, and the resulting unplanned vaporization.
For example, water at 10°C will flash into vapor and form bubbles at locations (such as the tip regions of impellers or suction sides of pumps) where the pressure drops below 1.23 kPa. The vapor bubbles (called cavitation bubbles since they form “cavities” in the liquid)
For example, water at 10°C will flash into vapor and form bubbles at locations (such as the tip regions of impellers or suction sides of pumps) where the pressure drops below 1.23 kPa. The vapor bubbles (called cavitation bubbles since they form “cavities” in the liquid)
pk = pv (for lequid type T)
Surface energy and surface tension, σ:
At boundaries between gas and liquid phases or between different immiscible liquids, molecular attraction introduces forces which cause the interface to behave like a membrane under tension.
Surface tension is an effect within the surface layer of a liquid that causes that layer to behave as an elastic sheet.
 |
| FIG 15 |
This effect allows insects (such as the water strider) to walk on water. It allows small metal objects such as needles, razor blades, or foil fragments to float on the surface of water, and causes capillary action.
Capillary Effect Another interesting consequence of surface tension is the capillary effect, which is the rise or fall of a liquid in a small-diameter tube inserted into the liquid. Such narrow tubes or confined flow channels are called capillaries. The rise of kerosene through a cotton wick inserted into the reservoir of a kerosene lamp is due to this effect. The capillary effect is also partially responsible for the rise of water to the top of tall trees. The curved free surface of a liquid in a capillary tube is called the meniscus.
 |
| FIG 16 |
units in Si : N/m
Equations of state
An equation of state is a thermodynamic equation describing the state of matter under a given set of physical conditions = Dependance between the fluid properties.
Liquids
The equations of state for most physical substances are complex and are expressible in simple forms only for limited ranges of conditions.
True for liquids as well
Gases
For gases in a highly superheated condition a useful aproximation is the theoretical equation of state for the perfect (ideal gas) - Clapeyron's equation:
P/ρ =RT
An ideal gas or perfect gas is a hypothetical gas consisting of identical particles of zero volume, with no intermolecular forces.
T - absolute temperature in K (or ⁰C); p - absolute pressure in N/m²; ρ - density in kg/m³;
R - gas constant in J/kgK (or J/kg⁰C) - for dry air R = 287 J/kg⁰C.
For ideal gas
Cp= specific heat at constant pressure;
Cv = specific heat at constant volume.
Change of state proceses for gasses
Isothermal process:
Constant pressure process:
Isentropic adiabatic process:
Zero heat transfer (adiabatic proces) and no friction (isentropic)
κ = Cp/Cv adiabatic constant for the gas.
Types Of Fluids Based ON Viscosity:
The fluids may be classified into following five types:
1. Ideal fluid
2. Real fluid
3. Newtonian fluid
4. Non-Newtonian fluid
5. Ideal plastic fluid
Types Of Fluids Based ON Density :
1- Compressible Fluid
2- Incompressible Fluid
3- Ideal plastic fluid
Ideal fluid:
A fluid which is incompressible and is having no viscosity, is known as ideal fluid. Ideal fluid is only an imaginary fluid as all the fluids which exists have some viscosity.
Real Fluids:
A fluid which possesses viscosity is known as real fluid. All the fluids in actual practice are real fluids.
Newtonian Fluids:
A real fluid in which the shear stress is directly proportional to rate of shear strain (or velocity gradient).
Non-Newtonian Fluid:
A real fluid in which the shear stress is not proportional to the rate of shear strain.
Ideal Plastic Fluid:
A fluid in which shear stress is more than the yield value and shear stress is proportional to the rate of shear strain (or velocity gradient).
 |
| FIG 17 |
Ideal fluid:
A fluid which is incompressible and is having no viscosity, is known as ideal fluid. Ideal fluid is only an imaginary fluid as all the fluids which exists have some viscosity.
Real Fluids:
A fluid which possesses viscosity is known as real fluid. All the fluids in actual practice are real fluids.
Newtonian Fluids:
A real fluid in which the shear stress is directly proportional to rate of shear strain (or velocity gradient).
Non-Newtonian Fluid:
A real fluid in which the shear stress is not proportional to the rate of shear strain.
Ideal Plastic Fluid:
A fluid in which shear stress is more than the yield value and shear stress is proportional to the rate of shear strain (or velocity gradient).
compressible fluid
A compressible fluid is a fluid in which significant density variations that occur during its flow have to be considered, as is usually the case with vapors and gases.
Incompressible fluid
Incompressible fluid means that the effects of pressure on the fluid density are zero or negligible. So the density and the specific volume of the fluid do not change during the flow.
Ideal Plastic Fluid
A fluid in which the shear stress is more than the yield value and the shear stress is directly proportional to the rate of shear strain that is it should follow newtons law of viscosity . It behaves as a rigid body at low stresses but flows as a viscous fluid at high stress .






















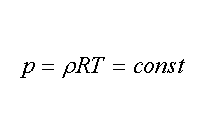




No comments:
Post a Comment
thanks for your visit